Key Components of Preventing Sample Contamination in Medical Diagnostic Labs: Training, Adherence to Guidelines, and Quality Control Measures
Summary
- Ensuring the accuracy of medical diagnostic Test Results is crucial for patient care in the United States.
- Protocols for preventing sample contamination in clinical labs are comprehensive and strictly enforced.
- Rigorous training, strict adherence to guidelines, and regular Quality Control measures are key components of preventing sample contamination in medical Diagnostic Labs.
Introduction
Accurate and reliable medical Diagnostic Tests are essential for physicians to make informed decisions about their patients' health. In the United States, clinical labs play a critical role in conducting these tests and providing accurate results. To ensure the validity of these results, strict protocols are in place to prevent sample contamination and maintain the integrity of the testing process.
Rigorous Training
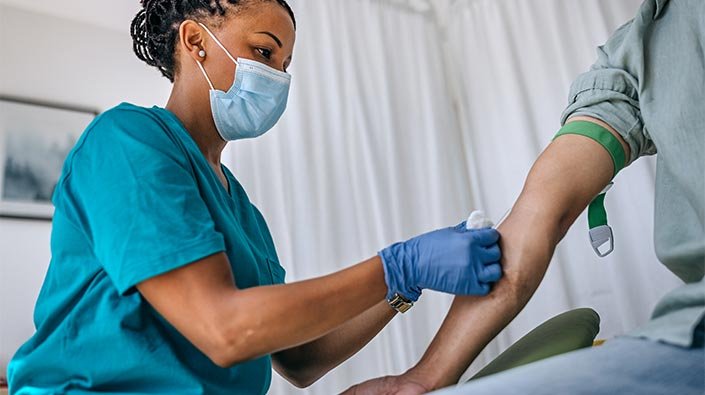
One of the key components of preventing sample contamination in medical Diagnostic Labs is ensuring that lab personnel receive rigorous training. This training covers a range of topics, including proper Sample Handling techniques, maintaining a sterile work environment, and following established protocols.
Proper Sample Handling Techniques
Lab technicians and other personnel are trained on how to properly handle samples to prevent contamination. This includes using sterile gloves, avoiding contact between samples, and properly sealing containers to prevent leaks or spills.
Maintaining a Sterile Work Environment
Keeping the work environment sterile is essential to prevent sample contamination. Lab personnel are trained on proper cleaning and disinfection procedures, including regular disinfection of surfaces and equipment, to minimize the risk of contamination.
Following Established Protocols
Strict adherence to established protocols is crucial for preventing sample contamination. Lab personnel are trained on the specific guidelines and procedures for each type of test they perform and are expected to follow these protocols to the letter.
Strict Adherence to Guidelines
In addition to rigorous training, strict adherence to guidelines is another important measure to prevent sample contamination in medical Diagnostic Labs. Guidelines are in place to ensure that all steps of the testing process are followed correctly and consistently.
Chain of Custody Procedures
Chain of custody procedures are used to track samples from the moment they are collected to the moment they are tested. This includes documenting who handled the sample, when and where it was collected, and any other relevant information to ensure the sample's integrity.
Sample Identification
Proper sample identification is essential to prevent mix-ups and contamination. Each sample must be clearly labeled with the patient's information and other relevant details to ensure that it is processed correctly.
Sample Storage
Proper sample storage is critical to prevent contamination. Samples must be stored at the correct temperature and humidity levels, and proper procedures must be followed to ensure that samples do not become compromised.
Regular Quality Control Measures
Regular Quality Control measures are essential to monitor the performance of medical Diagnostic Tests and ensure the accuracy of results. These measures help identify any issues or anomalies that could indicate sample contamination.
Internal Quality Control
Internal Quality Control measures are conducted within the lab to monitor the accuracy and precision of Test Results. This includes running control samples alongside patient samples to ensure that the testing process is working as intended.
External Quality Control
External Quality Control measures involve sending samples to external organizations for testing to validate the lab's results. This provides an independent assessment of the lab's performance and helps identify any potential issues with sample contamination.
Regular Audits and Inspections
Regular audits and inspections are conducted to ensure that the lab is following established protocols and guidelines. These audits help identify any areas for improvement and ensure that the lab is maintaining high standards of quality and safety.
Conclusion
Preventing sample contamination in medical Diagnostic Labs is crucial for ensuring the accuracy and reliability of Test Results. Rigorous training, strict adherence to guidelines, and regular Quality Control measures are key components of preventing sample contamination in clinical labs in the United States. By following these protocols, labs can provide accurate and reliable Test Results that are essential for patient care.
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on phlebotomy practices and healthcare. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
